Diagnostic Biomarker
Accurately, and Cost-Effectively, Detect Alzheimer’s Disease before Neurological Damage
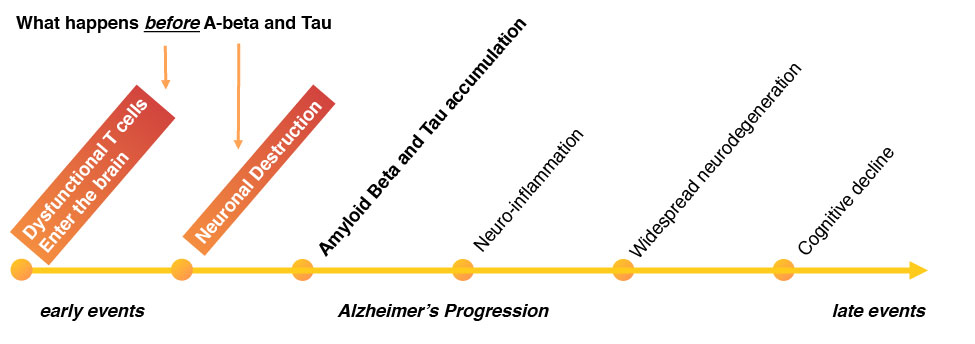
Data from the laboratory of Dr. Christopher Wheeler at Cedars-Sinai has identified one of the earliest events in the pathology of Alzheimer’s Disease. His data indicate that the expansion of certain aberrant T cells in the blood, and their subsequent entry into the brain, are among the earliest events in AD. These T cells deplete neurons in areas of the brain involved in AD. Dr. Wheeler has shown that these entry of these T cells into the brain leads to Ab and Tau deposition, as well as neuroinflammation and profound cognitive decline with age – the pathophysiological hallmarks of AD.
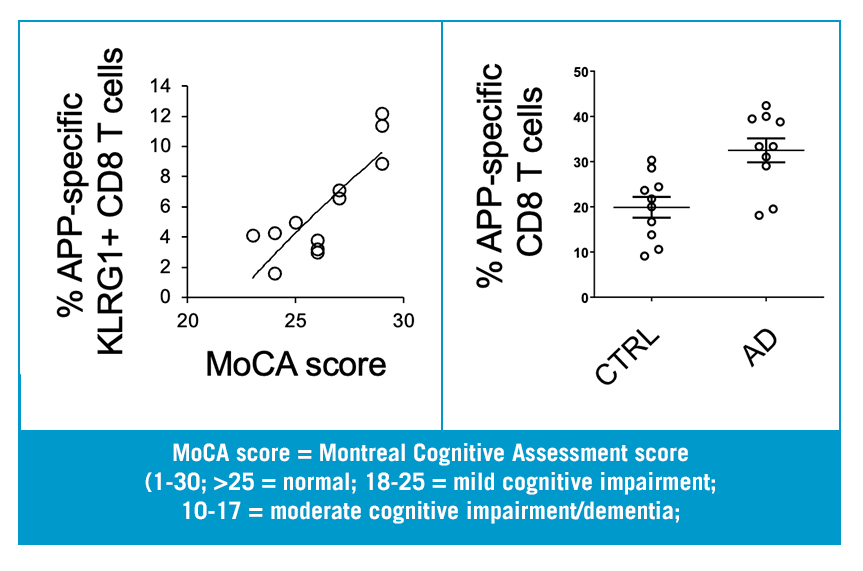
Dr. Wheeler has demonstrated that analogous T cells can be detected in human blood samples and their levels are highly correlated with Alzheimer’s Disease. He has devised a method to identify these T cells in human blood- the basis of the Diagnostic Biomarker Assay. Preliminary testing of this the Diagnostic Biomarker shows that it uniquely distinguishes early AD patients with little to no overlap between AD and control groups.
Additionally, levels of Diagnostic Biomarker in the blood correlate with cognitive decline (as determined by MoCA score).
Diagnostic Biomarker Workflow
Blood drawn
Blood put through a specialized biomarker kit tracker, time to incubate
Placed in flow cytometer to read information
Results determine pathologic TRM in blood
(“Diagnostic Biomarker”)
Presence of
Diagnostic Biomarker will Rule In/Rule Out AD diagnosis
T-TreatTM Therapeutic Intervention
In addition to their potential as a biomarker, Dr. Wheeler has shown that these abnormal T cells can be blocked from entering the brain in mice. This blockage prevents the characteristic pathophysiology of AD, including Ab and Tau deposition, as well as neuroinflammation. Importantly, blocking the T cells from entering the brain protects against cognitive decline that portends Alzheimer’s Disease. Dr. Wheeler’s method of blocking entry into the brain is the basis of the T-Treat Therapeutic Intervention.
"Novel Blood Cell Biomarker for Late Onset Alzheimer's Disease," Issued 1/11/2023
"Methods and Systems for Identification and Treatment of Pathological Neurodegeneration and Age-Related Cognitive Decline" Filed 2/13/2018